September 21, 2025
Better Therapy Selection for Childhood Leukemia
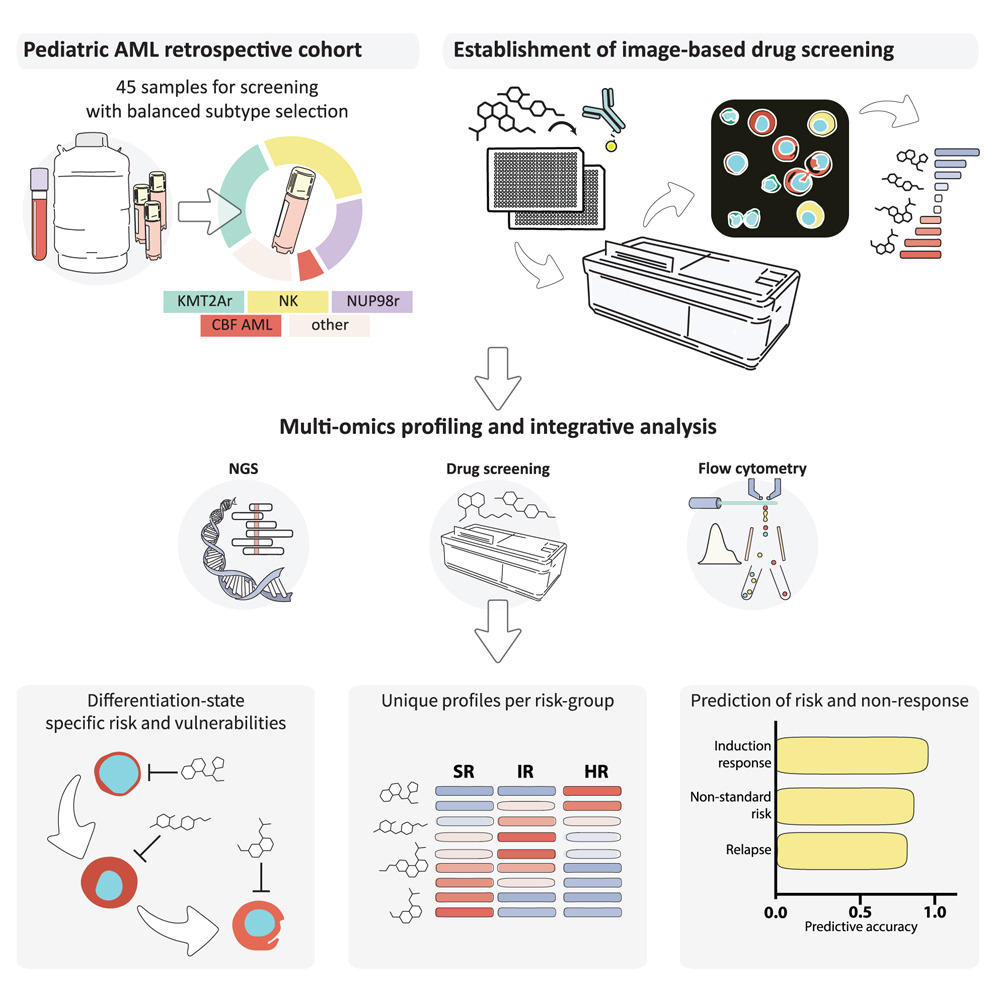
Despite decades of optimization of treatment protocols, the prognosis for acute myeloid leukemia in children (pediatric AML, pedAML) remains poor for many patients. Through a collaboration between CeMM, St. Anna Children’s Cancer Research Institute (CCRI), the Medical University of Vienna, and St. Anna Children’s Hospital we have now succeeded in developing a method for the early detection of resistance mechanisms in pediatric AML.
Pediatric acute myeloid leukemia (pediatric AML) is one of the most aggressive cancers in children. Unlike acute lymphoblastic leukemia, which occurs more frequently in children, pediatric AML is biologically more heterogeneous and in part more difficult to treat. The prognosis for many patients remains unsatisfactory: some do not respond to standard therapies or suffer relapses. Here we show that functional imaging and molecular characterization can be combined into a tool that detects therapy resistance already at diagnosis.
First author Ben Haladik, a PhD student in Kaan Boztug’s research group, together with the team further developed a platform for testing drug activity. It is based on the Pharmacoscopy method developed at CeMM for high-resolution imaging, combined with artificial intelligence and comprehensive molecular analysis. Using 45 patient samples, they demonstrated that robust predictions of therapy response and relapse risk can be derived.
Molecular Profile as a Key to Prognosis
Clear differences emerged between risk groups and even among subpopulations of cells that escape standard therapy. Particularly striking was a stem-cell-like form of leukemia that proved insensitive to conventional chemotherapy but was vulnerable to new combinations of known agents such as BCL2 and MDM2 inhibitors or HDAC blockers. The results show that functional analyses of this kind can further improve therapy prognosis for pediatric AML. While mutations provide important clues, the real clinical relevance lies in how leukemia cells respond to drugs.
This is precisely where the new method comes in: it makes the functional level visible and allows a direct link between molecular profile and actual therapy response.
From Research to the Clinic
First author Ben Haladik explains the methodology: “We have created a connection between molecular biological analyses, bioinformatics methods, and artificial intelligence that should provide a basis for further research toward better treatment options.”
Senior and corresponding author Kaan Boztug sees the study as a societal mission for the future: “Our study is the first to show that such ex-vivo drug tests can help us identify, at an early stage, patients whose leukemia cells are particularly resistant to standard therapy. Especially for these patients, we can then use the method to find targeted therapy options for pediatric AML. With our study we also position ourselves in a previously little-noticed field—the application of AI in childhood cancer research—as a significant player in European pediatric cancer research.”
A Milestone in Precision Medicine
“At CeMM we developed Pharmacoscopy, an imaging-based approach for functional single-cell precision medicine—a technology that enables true personalized medicine in cancer treatment. In the present study, this technology was further developed and successfully tested for the first time in the clinical diagnosis of pediatric AML. This is an important milestone toward implementing such methods on a larger scale for the benefit of pediatric patients,” says Giulio Superti-Furga, co-senior author of the study.
The study “Image-based drug screening combined with molecular profiling identifies signatures and drivers of therapy resistance in pediatric AML” was published in Cell Reports Medicine. DOI: 10.1016/j.xcrm.2025.102304
The authors: Ben Haladik, Margarita Maurer-Granofszky, Peter Zoescher, Raul Jimenez-Heredia, Alexandra Frohne, Anna Segarra-Roca, Chloe Casey, Felix Kartnig, Sarah Giuliani, Christina Rashkova, Peter Repiscak, Michael N. Dworzak, Giulio Superti-Furga, Kaan Boztug.
June 24, 2025
How a Faulty Transport Protein in the Brain Can Trigger Severe Epilepsy
read the full studies in Molecular Systems Biology
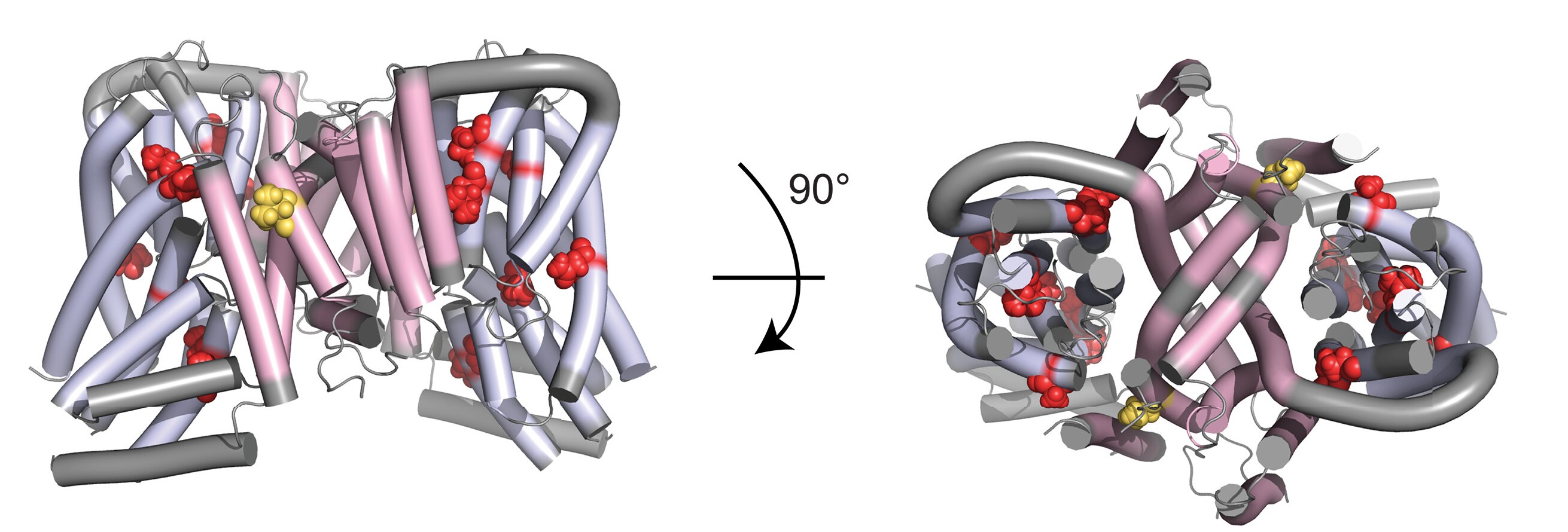
Citrate is essential for the metabolism and development of neurons. A membrane transport protein called SLC13A5 plays a central role in this process and has previously been linked to a particularly severe form of epileptic encephalopathy. This study built on data from the recently completed RESOLUTE and REsolution flagship projects and comprehensively investigates the function and structure of the membrane transporter SLC13A5, experimentally investigating 38 mutant variants.
In neurons, citrate plays an especially important role. As a so-called “neuromodulator,” it influences neuronal activity and is therefore present in relatively high concentrations in the cerebrospinal fluid. Accordingly, neurons express high levels of the SLC13A5 transporter to facilitate citrate uptake. When this transporter is not fully functional, it can lead to SLC13A5 Citrate Transporter Disorder—a severe form of epilepsy associated with impaired brain development (scientifically referred to as developmental epileptic encephalopathy, DEE). This condition is caused by mutations in the SLC13A5 gene.
Ten Thousand Mutations Analyzed
Here we used “deep mutational scanning” (DMS), analyzing the effect of nearly ten thousand different genetic mutations on the function of the SLC13A5 transport protein. The dataset was further enriched by computational analyses of protein stability, and 38 mutated SLC13A5 variants were selected for experimental investigation. We established differences in transporter production levels in neurons, their precise localization in the cell membrane, and the actual rate of citrate transport.
“With these results, we were able to identify and characterize disease-causing variants of the SLC13A5 transporter,” explains co-first author Jennifer Wen-An Wang, summarizing the main findings of the study. “In addition, by computationally analyzing the mutant variants, we assessed protein stability across different conformations and established an evolutionary conservation score for all variants,” adds co-first author Evandro Ferrada, now at the University of Valparaíso in Chile.
“Our work highlights the importance of systematically investigating the effects of genetic variants. Especially in rare diseases such as SLC13A5 citrate transporter deficiency, a specific form of epilepsy, this approach helps us uncover molecular disease mechanisms,” emphasizes senior author Giulio Superti-Furga. “At the same time, we gain valuable insights into the impact of variants that also occur in the general population – an important step toward a more comprehensive understanding of genetic diversity and its impact on human health.”
Patient data were provided by the TESS Research Foundation, which is dedicated to advancing research on SLC13A5 citrate transporter deficiency.
The Study “Large-scale experimental assessment of variant effects on the structure and function of the citrate transporter SLC13A5” was published in Science Advances on June 27, 2025. DOI: 10.1126/sciadv.adx3011
Authors: Wen-An Wang, Evandro Ferrada, Christoph Klimek, Tanja Osthushenrich, Aidan MacNamara, Tabea Wiedmer, Giulio Superti-Furga
June 2, 2025
Charting the human solute carrier transportome
read the full studies in Molecular Systems Biology:
Metabolic mapping of the human solute carrier superfamily
The genetic interaction map of the human solute carrier superfamily
The solute carrier superfamily interactome
Data- and knowledge-derived functional landscape of human solute carriers

An unprecedented international effort to decode how cells manage the transport of chemical substances has culminated in four groundbreaking studies published in Molecular Systems Biology. Led by Giulio Superti-Furga at CeMM and supported by an international consortium of academic and pharmaceutical partners under the European Union's Innovative Medicines Initiative, this decade-long project provides the first comprehensive functional blueprint of chemical transport pathways in human cells.
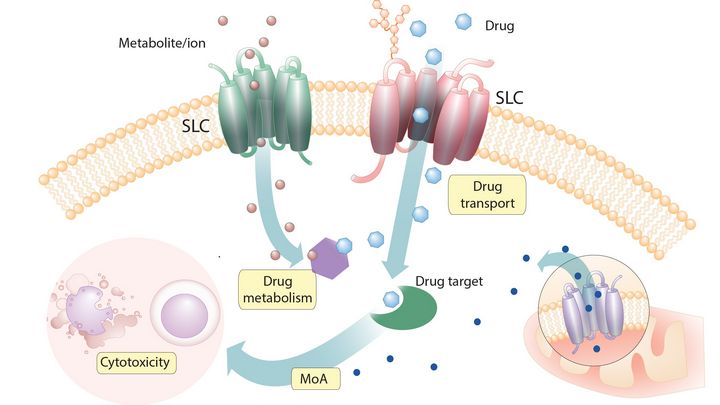
All life depends on the ability of cells to exchange substances with their environment. Nutrients, ions, and vitamins must be absorbed, while waste and special metabolites must be expelled. This fundamental process relies on transporter proteins embedded in cell membranes. Despite their crucial role, the function of many of the hundreds of human transporter-encoding genes has remained a mystery, slowing progress in fields ranging from cancer therapy to metabolic and neurological disease research.
Recognizing this challenge, the Superti-Furga group at CeMM first called for intensified research in a landmark 2015 article in Cell (https://doi.org/10.1016/j.cell.2015.07.022). Ten years later, CeMM and an international consortium have succeeded: by focusing on the largest family of transporters — the solute carriers (SLCs) — they have more than doubled existing knowledge and laid the foundations for future breakthroughs in understanding and targeting these critical proteins.
In a massive, coordinated effort, 120 researchers from 13 institutions across eight countries came together under the RESOLUTE consortium. After more than five years of laboratory work, most of the experimental data were gathered. The CeMM team, supported by a few key partners and primarily financed by the Austrian Academy of Sciences, then spent an additional year harmonizing, integrating, and interpreting the extensive datasets.
The result is a transformative expansion of the known biology of SLC transporters, integrating multidisciplinary experimental and computational approaches. Their findings illuminate the intricate logistics of chemical traffic within human cells, offering powerful new resources for the scientific and medical communities worldwide.
The effort not only generated vast scientific insights but also produced an impressive arsenal of reagents, datasets, and analytical tools — all made freely available to the global scientific community via https://re-solute.eu.
Funding: This major initiative was funded by the Innovative Medicines Initiative 2 Joint Undertaking under the European Union’s Horizon 2020 research and innovation program and the European Federation of Pharmaceutical Industries and Associations (EFPIA). Additional support was provided by the Austrian Academy of Sciences and participating institutions.
The four studies were published in Molecular Systems Biology:
- Metabolic mapping of the human solute carrier superfamily DOI: 10.1038/s44320-025-00106-4
- The genetic interaction map of the human solute carrier superfamily DOI: 10.1038/s44320-025-00105-5
- The solute carrier superfamily interactome DOI: 10.1038/s44320-025-00109-1
- Data- and knowledge-derived functional landscape of human solute carriers DOI: 10.1038/s44320-025-00108-2
January 30, 2025
Human genetic variants in SLC39A8 impact uptake and steady-state metal levels within the cell
read the full study in Life Sci Alliance
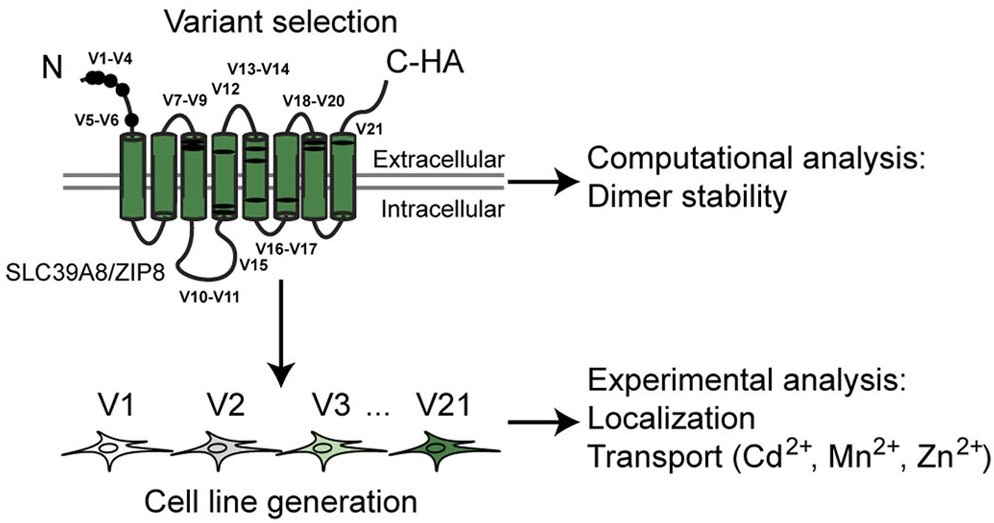
In another study from the collaborative projects RESOLUTE and REsolution we characterized 21 variants of hSLC39A8 (ZIP8), a metal transporter. Revealing diverse impacts on localization, ion uptake & stability—shedding light on disease mechanisms.
The focus was the human SLC39A8 gene, which encodes the metal-ion transporter protein ZIP8 responsible for the uptake of essential ions such as cadmium, manganese, zinc, iron, cobalt, and selenite. Mutations in this gene have been implicated in severe disorders like congenital disorder of glycosylation type II and Leigh-like syndrome, while many other variants remain of uncertain clinical significance. The research aimed to determine how 21 different SLC39A8 variants affect the localization and function of the ZIP8 protein.
We found that among the 21 variants, four completely disrupted protein expression, and five resulted in ZIP8 being largely retained in the endoplasmic reticulum, indicating issues with proper folding or trafficking. The remaining 12 variants correctly reached the plasma membrane; however, within this group, three variants exhibited a complete loss of metal ion transport activity. Moreover, five variants showed differential uptake efficiencies between metal ions, suggesting that even minor alterations in ZIP8 can significantly influence its transport function. Computational analyses further indicated that some mutations might destabilize the protein’s homodimer interfaces, potentially impairing overall structural stability and function.
In summary, the study highlights a range of effects that different SLC39A8 variants have on ZIP8, from impairing expression and trafficking to altering ion transport capabilities. These insights advance our understanding of how genetic variations in metal ion transporters contribute to disorders associated with disrupted metal homeostasis, emphasizing the importance of proper protein folding and dimerization in maintaining cellular function.
The study “Human genetic variants in SLC39A8 impact uptake and steady-state metal levels within the cell” was published in Life Sci Alliance on 30 January 2025, DOI: 10.26508/lsa.202403028.
Authors: Wen-An Wang, Andrea Garofoli, Evandro Ferrada, Christoph Klimek, Barbara Steurer, Alvaro Ingles-Prieto, Tanja Osthushenrich, Aidan MacNamara, Anders Malarstig, Tabea Wiedmer, Giulio Superti-Furga
January 3, 2025
The efflux pump ABCC1/MRP1 constitutively restricts PROTAC sensitivity in cancer cells
read the full study in Cell Chemical Biology
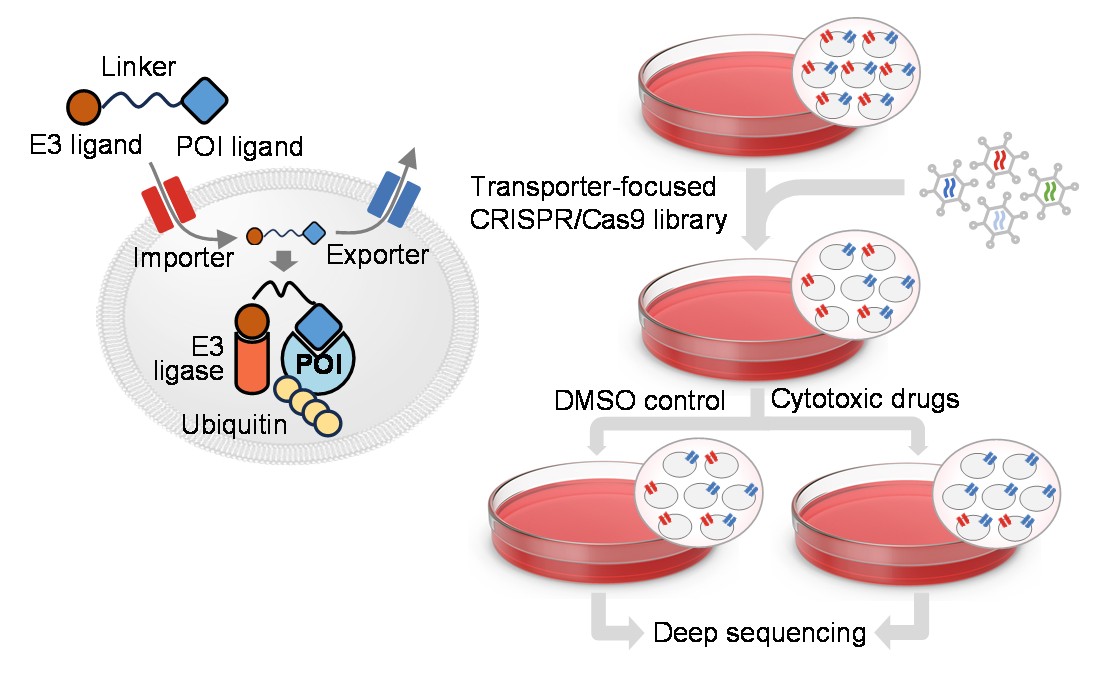
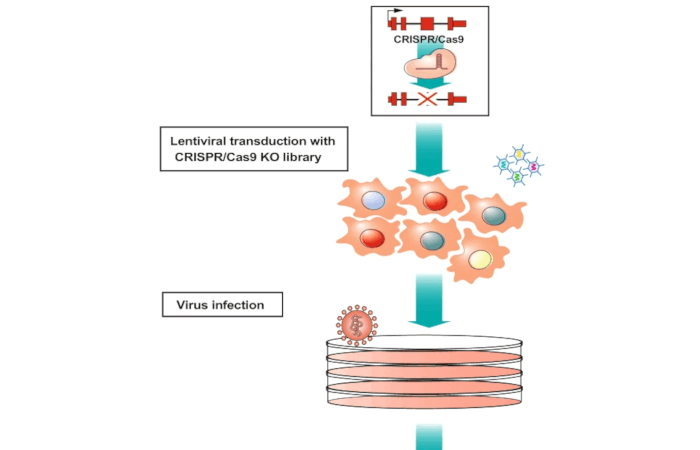
The research group of Giulio Superti-Furga at CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences has, in collaboration with the group of Alessio Ciulli, School of Life Sciences, University of Dundee, UK, used transporter-focused genetic screens and identified the ATP binding cassette transporter ABCC1/MRP1 as a key PROTAC efflux pump resistance factor.
The collaborative study was published in Cell Chemical Biology.
Transporter proteins such as the solute carrier (SLC) and ATP binding cassette (ABC) transporter superfamilies, play a crucial role in the movement of substrates such as nutrients, toxins and drugs across the cell membrane. These proteins are fundamental to cellular metabolism and are linked to various health conditions. Despite their importance and potential as therapeutic targets, many transporter proteins remain underexplored in pharmacological research. The research team led by Giulio Superti-Furga, Scientific Director at CeMM and professor at the Medical University of Vienna, is addressing this gap.
Most approved and experimental drugs target intracellular molecules, requiring them to cross the plasma membrane to be effective. Additionally, to achieve their desired therapeutic impact, these drugs must be resistant to cellular export mechanisms. Since the plasma membrane is impermeable to large molecules like drugs, transmembrane transporters are critical in facilitating both drug entry into cells as well as drug efflux. These transporters are thus important genetic factors that can influence the effectiveness of drugs.
The Superti-Furga group has investigated transmembrane transporters in more detail for over a decade now, having successfully identified several small molecule drug importers through SLC-focused genetic screens. In collaboration with Alessio Culli's team at the University of Dundee, this endeavor was extended by developing a new transporter-focused CRISPR library that includes three major groups of transmembrane transporters (451 SLCs, 48 ABC transporters and 40 P-Type ATPases) for use in genetic PROTAC resistance screens. PROTACs (proteolysis-targeting chimeras) are a class of bifunctional molecules designed to target specific proteins for degradation by recruiting them to the cell's built-in degradation system, thereby eliminating the protein rather than just inhibiting its function, like most small molecule drugs do.
This study revealed that the ABC transporter ABCC1, also known as multi drug resistance protein 1 (MRP1), plays a central role as the primary PROTAC efflux pump, increasing PROTAC resistance in human cancer cells up to 500-fold.
Giulio Superti-Furga, lead author on the paper details: “This finding is important because inhibiting ABCB1, another recently identified PROTAC transporter, may not prevent PROTAC resistance in cancers where ABCC1 is present. While it's uncertain if ABCC1 inhibition will be a viable strategy, we believe ABCC1 holds significant potential as a biomarker for PROTAC therapies.”
First author and senior scientist Gernot Wolf adds: “We believe the key impact of our work lies not just in identifying this universally expressed PROTAC exporter, but also in our approach, which could be applied to discover importers and exporters for any PROTAC or drug with a selectable phenotype. To also enable other research groups to study transporters using functional genetic screens, we made our genetic tools available to the research community through a non-profit repository.”
The study “The efflux pump ABCC1/MRP1 constitutively restricts PROTAC sensitivity in cancer cells” was published in Cell Chemical Biology on 3 January 2025, DOI: 10.1016/j.chembiol.2024.11.009.
Authors: Gernot Wolf, Conner Craigon, Shao Thing Teoh, Patrick Essletzbichler, Svenja Onstein, Diane Cassidy, Esther C.H. Uijttewaal, Vojtech Dvorak, Yuting Cao, Ariel Bensimon, Ulrich Elling, Alessio Ciulli, Giulio Superti-Furga
December 26, 2024
Ex vivo imaging-based high content phenotyping of patients with rheumatoid arthritis
read the full study in eBioMedicine

Researchers at CeMM and the Medical University of Vienna have developed an innovative microscopy-based approach that could predict the best approved drug to treat individual rheumatoid arthritis patients using samples of their blood. Published in the Lancet group journal EBioMedicine, the study demonstrates the feasibility of precision medicine to significantly enhance treatments for rheumatoid arthritis and likely other autoimmune diseases.
Rheumatoid arthritis is the most common inflammatory joint disorder, affecting over 60,000 people in Austria alone, with women being three times as likely to suffer from the condition as men. While treatment advances over the past decades have led to the development of numerous drugs with varying mechanisms of action, many patients still fail to achieve clinical remission due to a lack of tools to help find the right treatment, leaving their symptoms inadequately controlled.
Clinicians are left with a “trial-and-error” approach to therapy, where one drug is tested after another. While some biomarkers exist to help predict treatment outcomes, they are not yet suitable for routine clinical use or require invasive procedures.
In a long-standing collaboration, the team led by Giulio Superti-Furga at CeMM and the Medical University of Vienna has, for the first time, tested a precision medicine method that could enable more targeted and accurate therapy selection for rheumatoid arthritis and likely other autoimmune diseases. The findings, published in EBioMedicine, represent a major step forward in this field.
The method is based on cutting-edge microscopy technology capable of generating and analyzing vast amounts of imaging data in a fully automated manner. Developed at CeMM under the name “Pharmacoscopy”1, 2, it enables the direct measurement of drug effects on a wide variety of individual immune cells—a task that would be far too labor-intensive to perform at this scale using conventional molecular biology techniques. Additionally, it allows the observation of drug effects without the need to elucidate the underlying molecular mechanisms.
In the current study, the microscopy method was combined with a so-called "ex vivo" stimulation process. Immune cells from blood samples from patients are treated outside the body (“ex vivo”) with medications used for rheumatoid arthritis, and the effects of the drugs on immune cells are analyzed microscopically.
Through this approach, the researchers could take a snapshot of the behavior of immune cells in various conditions allowing for the identification of so-called "cellular phenotypes," that correlate with disease activity and therapeutic response. These phenotypes could potentially be used in the future to predict the success of different treatments on a blood sample before administering them to patients.
“The presented work is the first showcase for the application of imaging-based ex vivo screening in combination with ex vivo drug treatment as an approach to rheumatic diseases. This forms the basis for the future development of novel assays for precision medicine and preferential treatment selection,” explains Felix Kartnig, a PhD student in the group of Giulio Superti-Furga at CeMM and first author of the study.
“The establishment of this tool and its proof of concept forms a formidable scientific basis for the evaluation of this technology in the context of envisioned clinical trials and therefore highlights the value of innovative translational research.” states Leonhard Heinz co-corresponding author and principal investigator at the Division of Rheumatology of the Medical University of Vienna.
“Our approach is a remarkable example for a precision medicine of the future,” says study leader Giulio Superti-Furga. “Once again, our long-standing collaboration with the Medical University of Vienna has proven fruitful. We demonstrated that systematic and fully automated analyses of drug effects on individual cells can provide valuable insights into the mechanisms of action in human therapies.”
The study “Ex vivo imaging-based high content phenotyping of patients with rheumatoid arthritis” was published in EBioMedicine on 26 December 2024, DOI: 10.1016/j.ebiom.2024.105522.
Authors: Felix Kartnig, Michael Bonelli, Ulrich Goldmann, Noemi Mészáros, Nikolaus Krall, Daniel Aletaha, Leonhard X. Heinz, Giulio Superti-Furga
November 22, 2024
Membrane transporters modulating the toxicity of arsenic, cadmium, and mercury in human cells
read the full study in Life Science Alliance

PhD student Ferdigg, together with colleagues from the GSF Lab published a paper titled “Membrane transporters modulating the toxicity of arsenic, cadmium, and mercury in human cells" in the journal Life Science Alliance. They were employing loss- and gain-of-function genetic screens to unravel the role of membrane transporters in controlling heavy metal toxicity in human cells. They found that MRP1/ABCC1 protects against arsenic and mercury, SLC39A14 and SLC30A1 sensitize for cadmium, and interestingly SLC30A1 modulates cadmium toxicity through zinc.
This paper discusses how certain proteins in human cells help manage the toxicity of heavy metals like arsenic, cadmium, and mercury. The team used CRISPR/Cas9 loss-of-function (LOF) genetic screens to identify specific transporters that either protect cells from these toxic metals or make them more sensitive. The ATP-binding cassette (ABC) transporter and multidrug resistance protein MRP1/ABCC1 helps protect against arsenic and mercury, while the Solute Carrier Transporters (SLC) SLC39A14 and SLC30A1, increase sensitivity to cadmium. Additionally, using a reporter cell line to monitor metal accumulation and a cDNA gain-of-function screen, they elucidated the role of SLC30A1 in controlling cadmium toxicity through the modulation of intracellular zinc levels. This comprehensive transporter-wide approach provided new insights into the complex mechanisms by which membrane transporters influence heavy metal toxicity in human cells. Hence, this research provides new insights into how our cells handle these harmful substances and could lead to better ways to protect against heavy metal poisoning.
The study “Membrane transporters modulating the toxicity of arsenic, cadmium, and mercury in human cells” was published in Life Science Alliance on 22 November 2024, DOI: 10.26508/lsa.202402866.
Authors: , , ,
20 September, 2024
Identification and Characterization of Novel Small-Molecule Enhancers of the CUL3LZTR1 E3 Ligase KRAS Complex
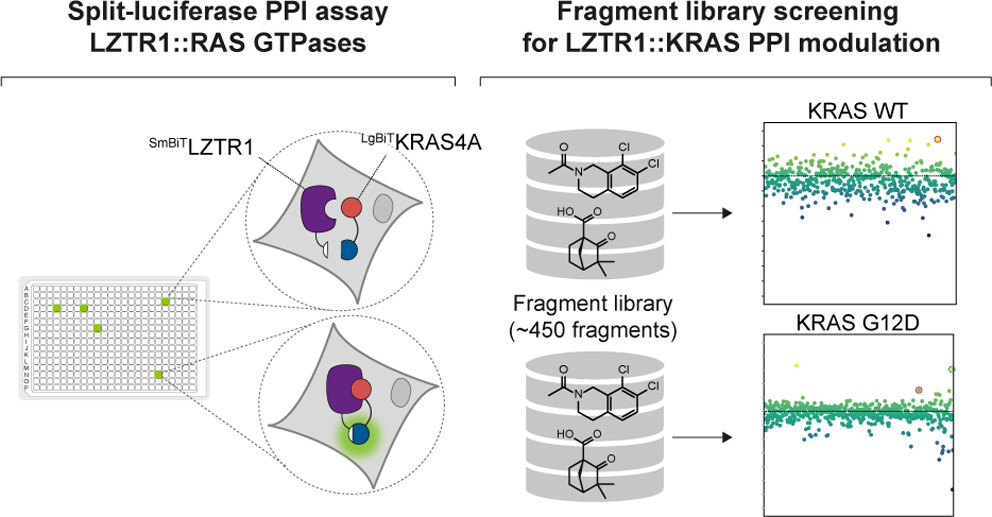
The GSF Lab’s research line on signaling in cancer published the finding of a new fragment engaging RAS which is capable of inducing association with LZTR1: Identification and Characterization of Novel Small-Molecule Enhancers of the CUL3LZTR1 E3 Ligase KRAS Complex.
The aim of the project of PhD student Sophie Piech, in collaboration with senior postdoc Johannes Bigenzahn, was to explore potential pharmacological strategies for modulating CUL3LZTR1-mediated KRAS degradation. The CUL3LZTR1 E3 ligase KRAS complex plays a crucial role in regulating cell growth and survival. By using advanced screening techniques, they identified several small molecules that can boost the function of this complex. These molecules have the potential to be developed into new cancer therapies, particularly for cancers driven by mutations in the KRAS gene
To that end the team developed a split-luciferase-based screening assay (SLA) to monitor the interaction between LZTR1 and different RAS GTPase substrates. Using that SLA setup, they were able to capture LZTR1 binding to its RAS GTPase family substrates KRAS, NRAS, HRAS as well as MRAS and RIT1. Furthermore, the assay was able to monitor mutational perturbations. Using this screening system, in combination with a small fragment library, they identified two fragments, C53 and Z86, that enhance interaction of the KRAS-LZTR1 complex in a dose-dependent manner. Finally, orthogonal validation experiments using proximity biotinylation (BioID), thermal shift assays, and NMR spectroscopy (in collaboration with the lab of Robert Konrat of University of Vienna) demonstrated fragment-dependent enhanced recruitment of endogenous LZTR1 and physical engagement of KRAS.
We think this study provides an exciting first step to screen for molecules harnessing the CUL3-LZTR1 E3 ligase complex and opens new avenues to target RAS signaling.
The study “Identification and Characterization of Novel Small-Molecule Enhancers of the CUL3LZTR1 E3 Ligase KRAS Complex” was published in ACS Chemical Biology on 20 September 2024, DOI: 10.1021/acschembio.4c00077.
Authors: Sophie Piech, Sven Brüschweiler, Josepha Westphalen, Katharina M. Siess, Julio García Murias, Robert Konrat, Johannes W. Bigenzahn, Giulio Superti-Furga
October 20, 2023
A conformation-locking inhibitor of SLC15A4 with TASL proteostatic anti-inflammatory activity

Scientists at CeMM, the Medical University of Vienna and the University of Lausanne succeeded for the first time in identifying and characterizing a new small molecule called "Feeblin", which can inhibit the interaction of the transporter protein SLC15A4 with the adapter protein TASL. Both proteins are part of proinflammatory signalling pathways in the body. In particular, patients with autoimmune diseases such as systemic lupus (SLE) could benefit from inhibiting the signalling pathway. The study has now been published in Nature Communications.
In autoimmune diseases, inflammation is chronic and leads to severe tissue damage. Several complex molecular pathways are involved in this process, but therapies and drugs targeting specific parts of these pathways remain scarce. Giulio Superti-Furga, Principal Investigator and Scientific Director of the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, in a previous study published in Nature, identified that a new adapter protein called TASL plays an essential role in signal transduction from the endo-lysosomal membrane transporter SLC15A4 and Toll-like receptors 7 and 9 -central players in innate immune defence- to the pro-inflammatory transcription factor IRF5 (interferon-regulatory factor 5). (Heinz et al, Nature 2020). From these findings, the group hypothesized that regulation of SLC15A4 and TASL might be an important aspect of immune signalling that could be therapeutically targeted to improve SLE. Giulio Superti-Furga and his team have now taken an important step towards achieving this goal and embarked on a drug discovery initiative that builds upon the group’s expertise in solute carriers and drug discovery.
For their study, the scientists developed a new test method that can be used to specifically monitor the presence of TASL. If TASL is not bound to SLC15A4, it is very unstable. Working with the CeMM Molecular Discovery Platform, Andras Boeszoermenyi, study author and postdoctoral fellow in Superti-Furga's laboratory, identified a small molecule that regulated the stability of TASL protein, and found that this was dependent on the presence of SLC15A4. The newly discovered compound called Feeblin – achieved exactly what the team hypothesized; the compound turned off pro-inflammatory signalling mediated by IRF5. The compound is named in honor of the Nobel Laureate Bruce Beutler's work on mutations in SLC15A4 signalling, the resultant mutant mouse strains were named "feeble. In a parallel study, the team of Maojun Yang at Tsinghua University elucidated the cryo-electron microscope structure of SLC15A4 and revealed that SLC15A4 undergoes major conformational changes when binding TASL. In collaboration with the team around Superti-Furga, the Yang laboratory confirmed the interaction of Feeblin with SLC15A4 and clarified its allosteric mechanism.
The abstract: Dysregulation of pathogen-recognition pathways of the innate immune system is associated with multiple autoimmune disorders. Due to the intricacies of the molecular network involved, the identification of pathway- and disease-specific therapeutics has been challenging. Using a phenotypic assay monitoring the degradation of the immune adapter TASL, we identify feeblin, a chemical entity which inhibits the nucleic acid-sensing TLR7/8 pathway activating IRF5 by disrupting the SLC15A4-TASL adapter module. A high-resolution cryo-EM structure of feeblin with SLC15A4 reveals that the inhibitor binds a lysosomal outward-open conformation incompatible with TASL binding on the cytoplasmic side, leading to degradation of TASL. This mechanism of action exploits a conformational switch and converts a target-binding event into proteostatic regulation of the effector protein TASL, interrupting the TLR7/8-IRF5 signaling pathway and preventing downstream proinflammatory responses. Considering that all components involved have been genetically associated with systemic lupus erythematosus and that feeblin blocks responses in disease-relevant human immune cells from patients, the study represents a proof-of-concept for the development of therapeutics against this disease.
The study “A conformation-locking inhibitor of SLC15A4 with TASL proteostatic anti-inflammatory activity” was published in Nature Communications on 20 October 2023, DOI: 10.1038/s41467-023-42070-3.
Authors: Andras Boeszoermenyi, Léa Bernaleau, Xudong Chen, Felix Kartnig, Min Xie, Haobo Zhang, Sensen Zhang, Maeva Delacrétaz, Anna Koren, Ann-Katrin Hopp, Vojtech Dvorak, Stefan Kubicek, Daniel Aletaha, Maojun Yang, Manuele Rebsamen, Leonhard X. Heinz, Giulio Superti-Furga

From 27 to 29 September 2023, the 'Unlocking Transporters for Drug Discovery' Conference took place at the historic Palais Niederösterreich in Vienna, Austria. The event, open for the first time to the entire scientific community, marked the conclusion of the RESOLUTE and REsolution projects, which are set to conclude by the end of 2023.
The REsolution and RESOLUTE consortia are public-private research partnerships supported by the Innovative Health Initiative (IHI), the European Union, and EFPIA. They are coordinated by CeMM and Pfizer and include partners from academia and the pharmaceutical industry. The RESOLUTE project aims to intensify global research on solute carrier (SLC) transporters and establish them as a novel target class for medical research, while the REsolution project aims to understand how genetic variants in humans affect the function of cellular SLCs.
Approximately 200 participants attended the scientific conference, which featured an engaging program including 45 top international scientists, with 20 external speakers from outside the consortia. The program also included two poster sessions with 60 abstract presenters. During the event, the RESOLUTE and REsolution consortia presented the results and lessons learned over the past five years to the broader scientific community. The talks covered various topics related to molecular transporters, their role in cell metabolism and regulation, structural biology, drug discovery, data integration, systems biology, as well as human physiology and disease.
The conference's theme was 'Fun Fair.' Participants had the opportunity to participate in various activities, including a virtual reality booth to explore the project's data, games organized by Nanion and Genscript, as well as a theater play featuring 'Transpy and Porty.'
We extend our gratitude to all participants for joining the event and engaging in meaningful scientific exchanges with other scientists and partners. Special thanks to Boehringer Ingelheim, Pfizer, and Sanofi for their support!
Learn more about the RESOLUTE Project: https://re-solute.eu/
July 28, 2023
Paralog-dependent isogenic cell assay cascade generates highly selective SLC16A3 inhibitors

The study’s first author Vojtech Dvorak, PhD student in Superti-Furga’s lab, adds, “One of the major obstacles for the development of novel drugs that target SLCs are the frequent functional redundancies among several transporters that are present in cells. This makes it very difficult to isolate the impact of a potential drug candidate on a single transporter and hence determine the selectivity. In one of our previous projects, we found a synthetic lethality between SLC16A1 and SLC16A3 present in several cell models. That is, the cell normally has both transporters, and when one of them is either inhibited by the drug, or the gene for one of them is lost, the other transporter can compensate. However, when say, the SLC16A1 gene is lost, the cell then actually depends on SLC16A3 for its survival (and vice versa). We realized that we could create several cell lines that are dependent on either of the distinct lactate transporters and use them to search for highly selective drugs.”
In the study published in Cell Chemical Biology, the scientists describe the development of the assay system, called Paralog-dependent isogenic cell assay, or PARADISO for short, and it’s use to develop a highly selective chemical probe targeting SLC16A3, called slCeMM1. Superti-Furga concludes, “The lack of specific cell-based assays is a problem for many promising drug targets, not only for SLCs. The logic of the PARADISO assay system should be in principle widely applicable and help to find new therapeutic targets.”
The abstract: Despite being considered druggable and attractive therapeutic targets, most of the solute carrier (SLC) membrane transporters remain pharmacologically underexploited. One of the reasons for this is a lack of reliable chemical screening assays, made difficult by functional redundancies among SLCs. In this study we leveraged synthetic lethality between the lactate transporters SLC16A1 and SLC16A3 in a screening strategy that we call paralog-dependent isogenic cell assay (PARADISO). The system involves five isogenic cell lines, each dependent on various paralog genes for survival/fitness, arranged in a screening cascade tuned for the identification of SLC16A3 inhibitors. We screened a diversity-oriented library of ∼90,000 compounds and further developed our hits into slCeMM1, a paralog-selective and potent SLC16A3 inhibitor. By implementing chemoproteomics, we showed that slCeMM1 is selective also at the proteome-wide level, thus fulfilling an important criterion for chemical probes. This study represents a framework for the development of specific cell-based drug discovery assays.
The study “Paralog-dependent isogenic cell assay cascade generates highly selective SLC16A3 inhibitors” was published on July 28, 2023, in Cell Chemical Biology (online ahead of print), doi: 10.1016/j.chembiol.2023.06.029
Authors: Vojtech Dvorak, Andrea Casiraghi, Claire Colas, Anna Koren, Tatjana Tomek, Fabian Offensperger, Andrea Rukavina, Gary Tin, Elisa Hahn, Sarah Dobner, Fabian Frommelt, Andras Boeszoermenyi, Viktoriia Bernada, J. Thomas Hannich, Gerhard F. Ecker, Georg E. Winter, Stefan Kubicek, Giulio Superti-Furga
February 1, 2023
A genome-wide CRISPR functional survey of the human phagocytosis molecular machinery
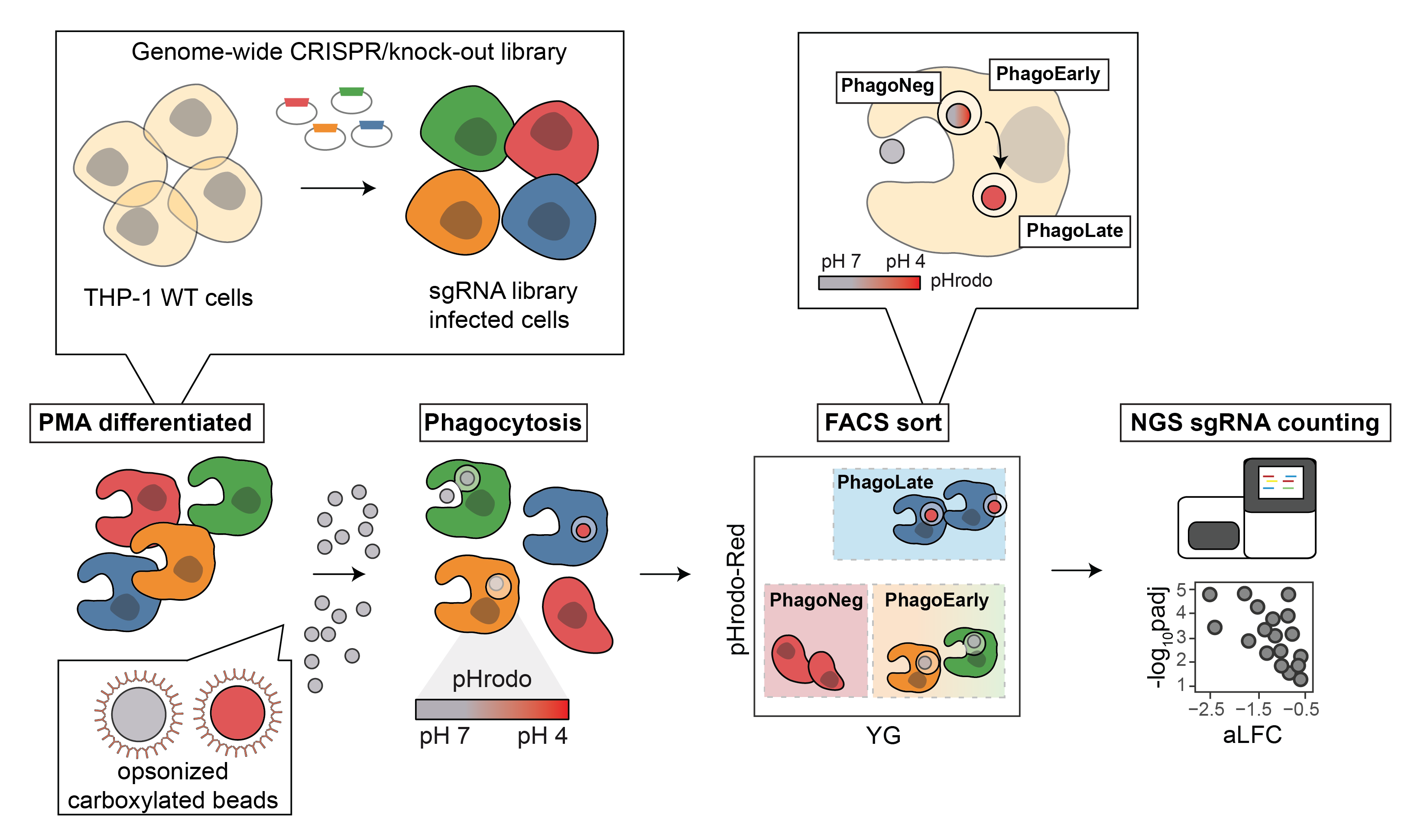
Based on unbiased genetic screens the GSF lab has in the past identified genes involved in a variety of cellular processes, including host-pathogen interactions (Sedlyarov et al., 2018), innate immunity and autoimmune diseases (Heinz et al., 2020), resistance to cytotoxic drugs (Girardi et al., 2020), chromatin state in dependence of metabolism (Li et al., 2021) and overcoming environmental nutrient restriction. Here we add to this list one of the, so we think, most comprehensive genetic map of phagocytosis regulators collected with a single reporter. We believe to have outlined the phagocytic machinery in an unprecedented number of components and can therefore offer an improved understanding of how cargo travels through the endo lysosomal system.
The abstract: Phagocytosis, the process by which cells engulf large particles, plays a vital role in driving tissue clearance and host defense. Its dysregulation is connected to autoimmunity, toxic accumulation of proteins, and increased risks for infections. Despite its importance, we lack full understanding of all molecular components involved in the process. To create a functional map in human cells, we performed a genome-wide CRISPRko FACS screen that identified 716 genes. Mapping those hits to a comprehensive protein–protein interaction network annotated for functional cellular processes allowed retrieval of protein complexes identified multiple times and detection of missing phagocytosis regulators. In addition to known components, such as the Arp2/3 complex, the vacuolar-ATPase-Rag machinery, and the Wave-2 complex, we identified and validated new phagocytosis-relevant functions, including the oligosaccharyltransferase complex (MAGT1/SLC58A1, DDOST, STT3B, and RPN2) and the hypusine pathway (eIF5A, DHPS, and DOHH). Overall, our phagocytosis network comprises elements of cargo uptake, shuffling, and biotransformation through the cell, providing a resource for the identification of potential novel drivers for diseases of the endo-lysosomal system. Our approach of integrating protein–protein interaction offers a broadly applicable way to functionally interpret genome-wide screens.
The study “A genome-wide CRISPR functional survey of the human phagocytosis molecular machinery” was published in Life Science Alliance on 1 February 2023. DOI:10.26508/lsa.202201715
Authors: Patrick Essletzbichler, Vitaly Sedlyarov, Fabian Frommelt, Didier Soulat, Leonhard X Heinz, Adrijana Stefanovic, Benedikt Neumayer and Giulio Superti-Furga.
Funding: CeMM and the Superti-Furga laboratory are supported by the Austrian Academy of Sciences. We further acknowledge receipt of third-party funds from the European Research Council (ERC) Advanced Grant 695214 GameofGates (P.E., V.S., G.S.-F.).
3rd REsolution and 7th RESOLUTE Consortia Meetings

From October 19 and 20-21, 2022, the consortia meetings of REsolution and RESOLUTE took place in Berlin. The event, hosted by Bayer, gathered 60 international participants. The REsolution and RESOLUTE consortia are public-private research partnerships supported by the Innovative Health Initiative (IHI), the European Union and EFPIA, coordinated by CeMM and Pfizer, and with partners from academia and the pharmaceutical industry. The RESOLUTE project aims at intensifying worldwide research on solute carrier (SLC) transporters and to establish them as a novel target class for medical research, while the REsolution project aims at understanding how genetic variants in humans affect the function of cellular SLCs.
On October 19, 2022, REsolution scientists worked in four groups to define the strategies for single-variant studies and SLC-family wide analyses. On October 20 and 21, 2022, the focus was on RESOLUTE scientific progress. In the last years, RESOLUTE has developed many biological resources and data sets focused on SLCs. In the last year of the project, it is the perfect time to use the project’s data sets in high-impact scientific articles. To that end, scientists presented the most promising subprojects, and they got key feedback to perform the final experiments. Another highlight of the event was the three keynote lectures presented by Melissa Miller (Pfizer), Michael Brands (Bayer) and Avner Schlessinger (Icahn School of Medicine at Mount Sinai). The final session was the RESOLUTE training activity: speed dating for participants to discuss a variety of topics one-to-one.
Thank you to all participants for joining the event and engaging in meaningful exchanges with all partners, and to special thanks to the speakers for their excellent presentations!
Scientific Symposium on the occasion of 15 Years of CeMM and 60th Birthday of Giulio Superti-Furga
On 20-21 May 2022, CeMM hosted the Scientific Anniversary Symposium on the occasion of the 15th anniversary of our institute and the 60th birthday of our Scientific Director. The conference on “Molecular medicine from signal transduction to societal impact: A journey through challenging territories” included an exciting line-up of renowned speakers working in the field of molecular biology across Europe and overseas. During the two-day event, 300 participants from the international scientific community, academia, industry, CeMM Faculty, colleagues and alumni, as well as other partner institutes and collaborators gathered at the House of Industry to discuss science, the development of new medical treatments and its impact on healthcare and society.
On the first day, MedUni Vienna Rector Markus Müller delivered the welcome address and congratulated CeMM Director Giulio Superti-Furga on his achievements, calling CeMM “an art form of itself”. Giulio Superti-Furga spoke a few introductory words and used the opportunity to thank important mentors, companions and colleagues for their support and for joining the event. Then the first session on “Decoding signals” kicked off with keynote speaker Tony Hunter, American Cancer Society Professor at the Salk Institute for Biological Studies (La Jolla, USA). During his presentation, Tony Hunter talked about histidine phosphorylation, a new prospect cancer target that could play a key role in the development of future medical therapies.
We hosted three additional sessions on understanding networks, harnessing proteostasis and drug development, and welcomed speakers from both, academia and industry. At the end of the afternoon, the guests came together in a cocktail reception and took the opportunity to interact, network, and discuss the scientific highlights as well as possible new collaborations in their research field.
On the second day, the program included four sessions with a focus on the societal aspects of research, with an outlook on how to create impact, and help patients, and highlighting what it takes to create context in challenging times such as a pandemic or the war.
Heinz Faßmann, President-elect of the Austrian Academy of Sciences delivered supportive and encouraging words about the future of CeMM, and the conference finished with the concluding remarks by CeMM Directors Giulio Superti-Furga and Anita Ender. Afterwards, participants joined a gala dinner in the stunning Palais Ferstel in the first district of Vienna. We are especially grateful to CeMM SAB Chair Carl-Henrik Heldin, who accompanied CeMM and its development from the very beginning, and livened up the evening with a very motivating dinner speech.
We would like to thank all speakers, participants, former and current CeMMies who joined us on this conference on the occasion of our institute's 15th anniversary and the 60th birthday of our Scientific Director. We are grateful for your engagement and for making it an extremely successful and pleasant event, packed with research highlights, great memories, new ideas and fruitful discussions!
Monica Bettencourt-Dias (IGC) and Giulio Superti-Furga (CeMM) appointed new chair and co-chair of EU-LIFE

Monica Bettencourt-Dias, Director of Instituto Gulbenkian de Ciência (IGC, Portugal), and Giulio Superti-Furga, Scientific Director of the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, have been appointed new Chair and Co-Chair respectively of EU-LIFE, the alliance of fifteen leading life science institutes in Europe, as of 1 January 2022.
During the next two years they will be working closely with Marta Agostinho, EU-LIFE Executive Director, to help shape the European research landscape, particularly with regard to excellent science. EU-LIFE’s priorities for the near future include the development of a 5-year strategy for the alliance and responding to the major challenges of our times, when research is more essential than ever, by reinforcing EU-LIFE action on the European policy agenda.
“We live in a time where science is taking centre stage in society and all of us know there are multiple challenges to face ahead, for scientific discovery, but also for the scientific endeavour itself: how science is done and how it relates to society. European scientists need to be drivers in shaping the future of Europe and EU-LIFE is a wonderful instrument for that, important for each individual institute that takes part in it, for the country it belongs to and for EUROPE. Couldn’t be more excited to take this role, together with my fellow directors, in particular co-chair Giulio Superti-Furga and EU-LIFE Executive Director Marta Agostinho” said Monica Bettencourt-Dias, Director of IGC and EU-LIFE new Chair.
“EU-LIFE has been key to me in learning and adopting best practices as Scientific Director of an institute devoted to research excellence. I am now delighted to start my role as EU-LIFE Co-Chair and to have the opportunity, with Chair Monica Bettencourt-Dias, to support even further its commitment to constant improvement. EU-LIFE’s member institutes aspire to create the best environment possible for research to flourish in all corners of Europe, and to have an impact in research culture beyond the alliance”, said Giulio Superti-Furga, Scientific Director of CeMM and EU-LIFE new Co-Chair.
The European Research Area and open science, Horizon Europe, widening excellence within Europe and synergies among national and European levels are some of the EU-LIFE focus areas. In addition, the alliance will continue to nurture its vibrant community in moving forward together on research governance and developing an ethical research culture that is open, collaborative and engaged with the society.
Monica Bettencourt-Dias was co-chair of EU-LIFE for the last two years and steps up as new chair of EU-LIFE, succeeding René Medema from The Netherlands Cancer Institute (NKI). We are very grateful to René for his outstanding term in leading EU-LIFE during such extraordinary times.
About EU-LIFE
EU-LIFE is an alliance of research centres whose mission is to support and strengthen European research excellence. EU-LIFE members are leading research institutes in their countries and internationally renowned for producing excellent research, widely transferring knowledge and nurturing talent. Since its foundation in 2013, EU-LIFE is a stakeholder in European policy participating regularly in the EU policy dialogue. More at: www.eu-life.eu
EU-LIFE Members
Centre for Genomic Regulation (CRG, Spain) | Central European Institute of Technology (CEITEC, Czech Republic) | European Institute of Oncology (IEO, Italy) | Flanders Institute For Biotechnology (VIB, Belgium) | Friedrich Miescher Institute for Biomedical Research (FMI, Switzerland) | Institut Curie (France) | Institute for Molecular Medicine Finland (FIMM, Finland) | Institute of Molecular Biology & Biotechnology (IMBB FORTH, Greece) I Instituto Gulbenkian de Ciência (IGC, Portugal) | International Institute of Molecular and Cell Biology in Warsaw (IIMCB, Poland) | Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC, Germany) | Research Center for Molecular Medicine of the Austrian Academy of Sciences (CeMM, Austria) | The Babraham Institute (Babraham, United Kingdom) | The Netherlands Cancer Institute (NKI, The Netherlands) | The University of Copenhagen Biotech Research & Innovation Centre (BRIC, Denmark).
December 14, 2021
Specific metabolic dependencies of cancer cells revealed by perturbation with tailored chemical library offer new therapeutic possibilities

Many types of cancer exhibit changes in their cellular metabolism. These contribute to the development and progression of cancer. Metabolic reprogramming has, thus, been recognized as a hallmark of cancer and may thus represent a vulnerability to be exploited by targeted cancer therapy. Scientists from the research group of Giulio Superti-Furga, Scientific Director at the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences and Professor at the Medical University of Vienna, have now used a drug library of 243 compounds targeting a variety of metabolic pathways to identify sensitivities among 15 myeloid leukemia cell lines. They were able to identify several specific pharmacological interventions possibilities.
In order to be able to grow and duplicate rapidly, proliferating cancer cells adapt their metabolism to meet the increased bioenergetic and biosynthetic demand. In fact, altered metabolism is considered a cancer hallmark. Cancer cells depend on this “high-powered” metabolic state to survive and grow, both in the body but also in cell culture. For some years now, the laboratory of Giulio Superti-Furga's at CeMM and MedUni Vienna has been working on understanding the dependency of specific functions in human cells on metabolites and nutrients. In a new study published in the journal Nature Communications, they report on the use of a small chemical compound library, called CLIMET, for CeMM Library of Metabolic Drugs, – for the purpose of experimentally testing which part of the altered metabolic program is most important, and thus critical, to different cancer cell types. The library consists of 243 active ingredients that influence the metabolism of cells by acting on different branches of the large intricate and widely connected network underlying cellular metabolism. The results highlight specific metabolic “vulnerabilities” of certain leukemia cell types, that may help conceive new therapeutic approaches.
Drug sensitivity provides important clues for therapeutic approaches
During her postdoc in the Superti-Furga laboratory at CeMM, first author Tea Pemovska developed the metabolic drug library, carefully selected for substances targeting individual pathways across the broad spectrum of metabolic processes operating in human cells. To get a better understanding of the molecular processes involved in cancer cell metabolism, the scientists performed a proof-of-concept survey using CLIMET on various blood cancer cell lines as well as patient samples. The obtained drug sensitivity profiles allowed the stratification of myeloid leukemia cell lines in five functional groups, each defined by differential sensitivity to 18 different compounds. Study leader Giulio Superti-Furga explains, "The collection of chemical agents that affect different aspects of cancer metabolism provides a toolkit to functionally assess cell lines, primary samples from cancer patients, and animal models in a versatile and dose-dependent way for their particular dependence on metabolic processes. Through this, we can get stratify cancer cell types not only by their molecular profile but by their actual metabolic needs. It is just a showcase but suggests a practical and actionable path towards an approach that exploits these cancer cell dependencies and vulnerabilities therapeutically, typically in combination with other drugs."
Identifying "drivers" and "vulnerabilities" of cell metabolism
Tea Pemovska, who is currently a scientist in the Functional Precision Hematology Group at MedUni Vienna, and colleagues, were able to show that certain human leukemia cell lines were particularly sensitive to the PI3K inhibitor Pictilisib, the fatty acid synthase inhibitor GSK2194069 and the SLC16A1 inhibitor AZD3965. She explains, "Some myeloid leukemia cell lines, especially two chronic myeloid leukemia cells, showed high selective sensitivity to the inhibitor AZD3965, which inhibits the important lactate transporter SLC16A1. This allows conclusions to be drawn about which cells and/or patients might best respond to this agent." At the same time, the study highlights the usefulness of a carefully assembled drug library with a metabolic focus to be used in phenotypic screening platforms, allowing the identification of metabolic dependencies. "Our study just delineates the feasibility of the strategy and emphasizes the importance of teasing out vulnerabilities of cancer cells by functional assays” says Giulio Superti-Furga.
The study “Metabolic drug survey highlights cancer cell dependencies and vulnerabilities” was published in the journal Nature Communications on 14 December 2021. DOI: 10.1038/s41467-021-27329-x.
Authors: Tea Pemovska, Johannes W. Bigenzahn, Ismet Srndic, Alexander Lercher, Andreas Bergthaler, Adrián César-Razquin, Felix Kartnig, Christoph Kornauth, Peter Valent, Philipp B. Staber and Giulio Superti-Furga;
Funding: The studywas supported by the European Research Council (695214 and 677006), the Austrian Science Fund (FWF SFB F4711), European Molecular Biology Organization (EMBO) Long Term Fellowship 733-2016, and the Vienna Science and Technology Fund (WWTF) through project LS16-034.

On 21 October 2021, the REsolution consortium held its first consortium meeting online with 30 participants. The REsolution consortium is a public-private research partnership supported by the Innovative Medicines Initiative (IMI), the European Union and the European Federation of Pharmaceutical Industries and Associations (EFPIA), with nine partners from academia and the pharmaceutical industry. With a duration of two years, the project aims at understanding how genetic variants in humans affect the function of cellular solute carrier transporters (SLCs).
Scientists from different work packages described the progress on the collection and annotation of human genetic variants for SLCs and associated phenotypes, the strategy to experimentally characterize selected mutations of prioritized SLCs, as well as the interpretation of variant effects based on structural information and compared to predicted effects by Variant Effect Predictors. Finally, participants discussed at the REsolution platform in GatherTown.
We would like to thank all participants for joining the event and their feedback, and specially the speakers for their excellent and informative presentations!
For more information on the project, please visit: http://re-solute.eu/resolution
September 06, 2021
Seitenwechsel candidate Alexandra Schmidl joins CeMM for a year
read about the Seitenwechsel Program

On 6 September 2021, schoolteacher Alexandra Schmidl joins CeMM as part of the Seitenwechsel program, which fosters fruitful exchanges among companies and teachers in Austria. During the next year, she will have a full immersion in the scientific environment at CeMM, the Research Center for Molecular Medicine of the Austrian Academy of Sciences, where she will benefit from daily interactions with CeMM researchers. The knowledge and new techniques that she acquires, will be then applied in her teaching when she goes back to her classroom next year. CeMM will not only profit from her everyday school life know-how, but will also gain further insights into the school management system in the second phase of the program.
With a background in biology and PhD in biology and botany, Seitenwechsel candidate Alexandra Schmidl is a biology teacher as well as physics teacher for younger pupils at the “GRG 21 Schulschiff Bertha von Suttner” (boat high school) in Vienna. During her time at CeMM, Alexandra will work together with the RESOLUTE project team, and will be involved in the generation of cell lines using CRISPR-Cas9, and the validation of protein reagents, i.e. antibodies, nanobodies, sybodies, for solute carriers (SLCs).
Seitenwechsel is a spin-off of the MEGA Education Foundation, which promotes innovative educational initiatives in the areas of chance/fairness and business competence/life skills to support Austrian talent. On this year’s edition, the Seitenwechsel project has selected eight committed and determined teachers who will take the opportunity to deepen their experience by working in a company for a year. On the second phase, company managers will also participate in an exchange by joining the teachers’ schools for a few days to learn more about the school management on the second year. Teachers will also bring impulses from their professional experience back into their schools and implement these new fresh approaches in their teaching to better prepare the pupils for a successful start into their own career.
Continuing education, gaining knowledge, lifelong curiosity and the desire to help shape the future are essential criteria for success, both in the researchers' and the teachers' profession. As a pioneering and leading international biomedical research institute in Austria, CeMM is committed to making society more resilient to future challenges - through facts, competence and the promotion of collaboration and diversity in science.
CeMM supports gladly the Seitenwechsel initiative, and is looking forward to an enriching exchange with Alexandra, who will also bring new fresh ideas and ways of seeing the scientific world from the educators’ perspective. We wish Alexandra a great start and a very successful year, full of learning, engagement and fulfilling experiences at CeMM!
August 10, 2021
An Overview of Cell-Based Assay Platforms for the Solute Carrier Family of Transporters
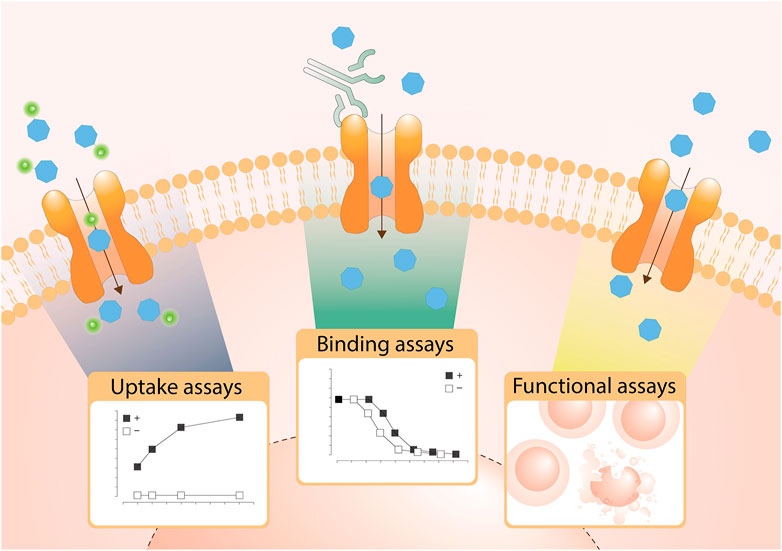
Are you Interested in assaying Solute Carriers?
The GSF lab and many members of the RESOLUTE consortium have, in a great team effort, composed a structured overview of cellular assay technologies for SLCs. This is definitely an important step towards providing the scientific community with novel approaches to functionally investigate SLCs. We also hope that this will guide many drug discovery-endeavors in the near future.
The abstract The solute carrier (SLC) superfamily represents the biggest family of transporters with important roles in health and disease. Despite being attractive and druggable targets, the majority of SLCs remains understudied. One major hurdle in research on SLCs is the lack of tools, such as cell-based assays to investigate their biological role and for drug discovery. Another challenge is the disperse and anecdotal information on assay strategies that are suitable for SLCs. This review provides a comprehensive overview of state-of-the-art cellular assay technologies for SLC research and discusses relevant SLC characteristics enabling the choice of an optimal assay technology. The Innovative Medicines Initiative consortium RESOLUTE intends to accelerate research on SLCs by providing the scientific community with high-quality reagents, assay technologies and data sets, and to ultimately unlock SLCs for drug discovery.
The study “An Overview of Cell-Based Assay Platforms for the Solute Carrier Family of Transporters” was published in Frontiers in Pharmacology on August 10, 2021, DOI: 10.3389/fphar.2021.722889.
Authors: Vojtech Dvorak, Tabea Wiedmer, Alvaro Ingles-Prieto, Patrick Altermatt, Helena Batoulis, Felix Bärenz, Eckhard Bender, Daniela Digles, Franz Dürrenberger, Laura H. Heitman, Adriaan P. IJzerman, Douglas B. Kell, Stefanie Kickinger, Daniel Körzö, Philipp Leippe, Thomas Licher, Vania Manolova, Riccardo Rizzetto, Francesca Sassone, Scarabottolo, Avner Schlessinger, Vanessa Schneider, Hubert J. Sijben, Anna-Lena Steck, Hanna Sundström, Sara Tremolada, Maria Wilhelm, Marina Wright Muelas, Diana Zindel, Claire M. Steppan and Giulio Superti-Furga
Funding: This work is part of the RESOLUTE project that has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under Grant Agreement No. 777372. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA. In addition, we acknowledge support by the Austrian Academy of Sciences, the Vienna Science and Technology Fund (WWTF, Grant No. LS17-051), Austrian Science Fund (FWF, Grant No. W1232), Biotechnology and Biological Sciences Research Council (BBSRC, Grant No. BB/P009042/1), Novo Nordisk Foundation (grant NNF20CC0035580), National Institutes of Health (NIH, Grant No. R01 GM108911), Bayer AG, Vifor Pharma, Sanofi and Pfizer Inc. The funders were not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication. All authors declare no other competing interests.
May 10, 2021
Cell-surface SLC nucleoside transporters and purine levels modulate BRD4-dependent chromatin states

Do purines influence cancer development?
Numerous disease development processes are linked to epigenetic modulation. One protein involved in the process of modulation and identified as an important cancer marker is BRD4. Our recent study shows that the supply of purines as well as the purine synthesis of a cell can influence BRD4 activity and thus play a role in the carcinogenesis process.
The protein BRD4, which makes a decisive contribution to the unpacking and packaging of DNA in chromatin, was identified as a marker for cancer. How can BRD4 be modulated? We investigated how certain externally supplied purines influence BRD4 and thus the development process of various cancer diseases. Disturbances of the purine metabolism in the cell have already been associated with some disease patterns in the past. We show, on the one hand, that inhibiting purine supply as well as disturbing purine synthesis can trigger a functional disturbance of BRD4 and thus impact chromatin accessibility. On the other hand, BRD4 functionality could be restored by adding adenine.
Analysis of the transport pathways
The group focuses particularly on those transport proteins in the genome that transport numerous important substances such as nutrients and metabolites into and out of the cell – so-called solute carriers (SLC). Kai-Chun Li explains: “Our aim was to investigate the involvement of SLC-mediated purine uptake and cellular metabolism in the modulation of cellular epigenetic states, because purine metabolism plays an essential role in cell metabolism.” With the help of SLCs, we were able to modulate the purine supply for their study and observe the direct effects. We used both a genetic screening based on a CRISPR/Cas9 library focused on transporters, and a drug screening using a compound library mainly consisting of cellular metabolites and drugs, both to track down the modulation of BRD4-dependent chromatin states in myeloid leukemia cells. We compared “normal” cancer cells with those cancer cells in which the SLCs that transport purines were inhibited. In addition, purines were added to or omitted from the growth medium of the cells in various experiments, thus modulating purine biosynthesis in the cells.
Adenine brings BRD4 back into balance
The study shows that an imbalance of intracellular purine pools leads to a dysfunction of BRD4-dependent transcriptional modulation of chromatin, which means that the correct reading of DNA information is disturbed. “These results demonstrate a pharmacologically effective axis between purine metabolism and BRD4-dependent chromatin states,” explains Giulio Superti-Furga. Drugs that influence BRD4 have already been developed in the past. At the same time, some cancer types also became resistant to such BET inhibitors. “With our study, we show another way to modulate BRD4 – by influencing the purine metabolism.” We also found an answer to the question of how BRD4 functionality could be restored: we were able to show that adenine, a purine-derived compound, plays a strong role in BRD4 interaction. “Our results suggest that adenylates (adenine, ATP, etc.) are important for healthy cells. This could be a significant starting point for developing new therapies against BRD4-induced cancer types,” says Superti-Furga.
The study “Cell-surface SLC nucleoside transporters and purine levels modulate BRD4-dependent chromatin states” was published in Nature Metabolism on May 10, 2021, DOI: 10.1038/s42255-021-00386-8.
Authors: Kai-Chun Li, Enrico Girardi, Felix Kartnig, Sarah Grosche, Tea Pemovska, Johannes W. Bigenzahn, Ulrich Goldmann, Vitaly Sedlyarov, Ariel Bensimon, Sandra Schick, Jung-Ming G. Lin, Bettina Gürtl, Daniela Reil, Kristaps Klavins, Stefan Kubicek, Sara Sdelci, Giulio Superti-Furga
Funding: We acknowledge receipt of third-party funds from the Austrian Science Fund (FWF SFB F4711, F.K., J.W.B., G.S-F.; I2192-B22 ERASE, V.S.), the European Research Council (ERC AdG 695214 GameofGates, K-C.L., E.G., A.B., U.G., G. S-F.), the European Commission (Marie Sk1odowska-Curie Action Fellowship 661491, E.G.) and an EMBO long-term Fellowship (ALTF 733-2016, T.P.). S.G. is supported by the Peter and Traudl Engelhorn Foundation. Research in the Kubicek lab is supported by the Austrian Science Fund (FWF F4701) and the European Research Council under the European Union’s Horizon 2020 research and innovation programme (ERC-CoG-772437).
December 1, 2020
SLC25A51 regulates the transport of the coenzyme NAD into the mitochondria

For their growth, cells need various nutrients and vitamins. So-called solute carriers (SLC), proteins that can transport such substances across the boundaries of cellular membranes, play a central role in metabolism. Scientists in Giulio Superti-Furga’s research group at the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences have now discovered that the previously uncharacterized protein SLC25A51 acts as a transporter into the mitochondria for the coenzyme NAD. This molecule has already been associated with numerous physiological and pathological processes such as ageing, neurological diseases and the metabolism of cancer cells. Therefore, the results of this study not only open up new possibilities to study the biological role of NAD but also potentially provide the basis for new therapeutic approaches. The work has now been published in the journal Nature Communications.
Solute carriers (SLC) are proteins that act as transporters and enable the entry and exit of nutrients and waste products into and from the cell and its organelles. Many of these transporter proteins are still relatively poorly studied and the question of how some nutrients enter and leave cells often remains unanswered. So far, it has not yet been clarified how mitochondria gain access to an important cofactor of our metabolism, the so-called NAD (nicotinamide adenine dinucleotide). In scientific literature, there were only references to mitochondrial NAD transporters in plants and yeast. Lead author Enrico Girardi and the research group of CeMM Scientific Director Giulio Superti-Furga, in cooperation with scientists from the University of Bari (Italy), have now identified the protein responsible for the important transport of NAD into mitochondria: SLC25A51.
The study "Epistasis-driven identification of SLC25A51 as a regulator of human mitochondrial NAD import" was published in Nature Communications on 1 December 2020. DOI: 10.1038/s41467-020-19871-x
Authors
Enrico Girardi, Gennaro Agrimi, Ulrich Goldmann, Giuseppe Fiume, Sabrina Lindinger, Vitaly Sedlyarov, Ismet Srndic, Bettina Gürtl, Benedikt Agerer, Felix Kartnig, Pasquale Scarcia, Maria Antonietta Di Noia, Eva Liñeiro, Manuele Rebsamen, Tabea Wiedmer, Andreas Bergthaler, Luigi Palmieri, Giulio Superti-Furga
Funding
The study was funded with support by the Austrian Academy of Sciences, the European Research Council (ERC) (AdG 695214, StG 677006) and the Austrian Science Fund (FWF P29250-B30, FWF DK W1212).
May 13, 2020
TASL is the SLC15A4-associated adaptor for IRF5 activation by TLR7–9

How cells recognize pathogens and alert the immune system is a fundamental process of high importance for the survival of any species, including humans. A key role is ascribed to so-called adapters, that equal little molecular platforms inside cells where signals from pathogen detectors are integrated for safety and accuracy and conveyed to lasting signals leading to the activation of the major “red alarm” genes, like interferons. Researchers from the lab of Giulio Superti-Furga in collaboration with Boehringer Ingelheim in Ridgefield (US) have identified a new key element of the multi-component machinery that is responsible for sorting out the nature and severity of the pathogen challenge. The new protein, named TASL, is indispensable for the signaling of so-called Toll-like receptors (TLR) in the endosomes leading to activation of the gene-activator IRF5 in certain immune cells. Sensitive “tuning” of the machinery is highly important as too much output causes inflammation also in the absence of the pathogen, as in auto-immune diseases. This particular version of the machinery seems particularly associated with disorders such as systemic lupus erythematosus (SLE). This discovery highlights a potential new target for the development of drugs to treat certain autoimmune diseases and possibly also overreaction to viral and other infections and has been published in the renowned scientific journal Nature.
Previous studies revealed that SLC15A4, a member of the body’s biggest family of transporter proteins, was known as an essential component required for the correct function of these TLRs. Based on their strong research interests in pathogen-sensing by the innate immune system and the characterization of solute carriers, researchers in the group of Giulio Superti-Furga set out to investigate how SLC15A4 influences the ability of TLRs to sense pathogens, and, consequently, gain a better understanding on its implication in autoimmune conditions, and in particular SLE.
In their study, first author Leonhard Heinz and the team, including Boehringer Ingelheim researchers, undertook a precise investigative work, not taking for granted previous findings on SLC15A4 and the connection to this group of specially located TLRs. They painstakingly determined by biochemistry and mass spectrometry the molecular interactions that involved SLC15A4. This led to the identification of an uncharacterized protein CXorf21, belonging to the functionally orphan genes that are merely numbered and assigned to the chromosome of origin. The gene, like SLC15A4, had been previously loosely associated with SLE.
The team demonstrated that the interaction between TASL and SLC15A4 was crucial for the localization and function of the TASL protein and could pinpoint the precise involved portions of both proteins. A eureka moment for the understanding of the protein came with the observation that TASL harbors a specific motif essential for the recruitment and activation of IRF5. “After STING, MAVS and TRIF, the new protein TASL is the fourth key innate immunity adaptor functioning as a platform for the encounter of a kinase and a gene activator of the IRF family”, says Manuele Rebsamen, CeMM senior postdoctoral fellow and project leader of the study.
This study is the result of a collaboration between CeMM and the Drug Concept Discovery Group led by Charles Whitehurst and JangEun Lee in the Immunology and Respiratory Diseases Department at Boehringer Ingelheim (Ridgefield, CT, USA). Researchers also benefited from the support of the Proteomics and Metabolomics (Pro-Met-) facility and the Biomedical Sequencing Facility (BSF) at CeMM.
The study “TASL is the SLC15A4-associated adaptor for IRF5 activation by TLR7–9” was published in Nature on 13 May 2020. DOI: 10.1038/s41586-020-2282-0.
Authors
Leonhard X. Heinz, JangEun Lee, Utkarsh Kapoor, Felix Kartnig, Vitaly Sedlyarov, Konstantinos Papakostas, Adrian César-Razquin, Patrick Essletzbichler, Ulrich Goldmann, Adrijana Stefanovic, Johannes W. Bigenzahn, Stefania Scorzoni, Mattia D. Pizzagalli, Ariel Bensimon, André C. Müller, F. James King, Jun Li, Enrico Girardi, M. Lamine Mbow, Charles E. Whitehurst, Manuele Rebsamen, Giulio Superti-Furga
Funding
The study was funded with support by the Austrian Academy of Sciences, the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (Grant Agreement No. 695214, awarded to Giulio Superti-Furga), the Austrian Science Fund (FWF SFB F4711) and by Boehringer Ingelheim (Research Collaboration Agreement BI-CeMM 238114).
March 09, 2020
A widespread role for SLC transmembrane transporters in resistance to cytotoxic drugs

Researchers around first author Enrico Girardi of the Giulio Superti-Furga lab at CeMM have studied how Solute Carriers (SLCs), a large family of membrane transport proteins, influence the activity and potency of cytotoxic drugs. The results show that many cytotoxic compounds need at least one functioning solute carrier transporter to unfold their activity. "The use of a custom-made, SLC-focused library was instrumental in allowing us to screen a large number of compounds, revealing hundreds of SLC-drug associations and providing many novel insights into SLC biology and drug mechanisms", says Enrico Girardi, CeMM senior postdoctoral fellow and first author of the study. Giulio Superti-Furga, CeMM Scientific Director and last author of the study adds: "This study raises strong doubts that the generally accepted idea that most drugs can enter cells by simply diffusing through its membrane is correct and highlights the increasingly appreciated need to systematically studying the biological roles of solute carriers". The present study is the result of a cross-disciplinary collaboration with researchers from the University of Vienna Pharmacoinformatics Research Group of Gerhard Ecker as well as the group of Stefan Kubicek at CeMM.
Authors
Enrico Girardi, Adrián César-Razquin, Sabrina Lindinger, Konstantinos Papakostas, Justyna Konecka, Jennifer Hemmerich, Stefanie Kickinger, Felix Kartnig, Bettina Gürtl, Kristaps Klavins, Vitaly Sedlyarov, Alvaro Ingles-Prieto, Giuseppe Fiume, Anna Koren, Charles-Hugues Lardeau, Richard Kumaran Kandasamy, Stefan Kubicek, Gerhard F. Ecker & Giulio Superti-Furga
Publication
Girardi, E., César-Razquin, A., Lindinger, S. et al.
A widespread role for SLC transmembrane transporters in resistance to cytotoxic drugs.
Nat Chem Biol (2020). https://doi.org/10.1038/s41589-020-0483-3
Funding
The study was funded with support by the Austrian Academy of Sciences, the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (Grant Agreement No. 695214, awarded to Giulio Superti-Furga), the Austrian Science Fund (FWF I2192-B22 ERASE; FWF P29250-B30 VITRA) and by a Marie Sklodowska-Curie fellowship to Enrico Girardi (MSCA-IF-2014-661491). Research in the Kubicek laboratory is supported by the Austrian Federal Ministry for Digital and Economic Affairs and the National Foundation for Research, Technology, and Development, the Austrian Science Fund (FWF) F4701 and the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (ERC-CoG-772437). The Pharmacoinformatics Research Group (Ecker lab) acknowledges funding provided by the Austrian Science Fund FWF AW012321 MolTag.
July 18, 2019
New insights into cellular response to oncolytic VSV infection
CeMM researchers from the laboratory of Giulio Superti-Furga, with Anna Moskovskich as first author of the study, have identified two transporters, SLC35A1 & SLC30A1, with opposite effects on the regulated cell death induced by the virus.
Special thanks to Enrico Girardi, Co-Supervisor of PhD Student Anna, and Gijs Versteeg (Universität Wien) and Michael Freissmuth (Medizinische Universität Wien) for critical feedback.
Publication
The transporters SLC35A1 and SLC30A1 play opposite roles in cell survival upon VSV virus infection.
Moskovskich A, Goldmann U, Kartnig F, Lindinger S, Konecka J, Fiume G, Girardi E, Superti-Furga G.
Sci Rep. 2019 Jul 18;9(1):10471. doi: 10.1038/s41598-019-46952-9.
Funding
The study was supported with competitive funds from the Austrian Science Fund, European Research Council and European Commission.

RESOLUTE, an European Union IMI2 funded project with 13 partners, CeMM as academic coordinator and Pfizer as EFPIA leader, celebrated the 2nd RESOLUTE consortium meeting from June 3-5, 2019 in Krems an der Donau, near Vienna.
More than 65 participants coming from all over Europe and the US joined the meeting. This was a great chance to review the current status of the RESOLUTE project right before completing the first year of collaborations. This edition featured many young investigators presenting their progress in the generation of reagents, data and assays for solute carriers. Additionally, strategies were developed for tackling the challenges of the second year of RESOLUTE. Participants as well enjoyed the excellent weather and cultural amenities of the beautiful vineyards of the Wachau area.
Learn more about the RESOLUTE Project: https://re-solute.eu/
November 15, 2018
New mechanism controlling the master cancer regulator uncovered

Who regulates the key regulator? The Superti-Furga laboratory at CeMM reports online in the journal Science about a newly discovered mechanism by which RAS proteins, central to cancer signaling, are regulated in their activity and localization.
Of the more than 23,000 genes in the human genome, only a handful assume a very central role in signal transduction and growth regulation. Of these, the three genes encoding RAS proteins are particularly important, as they are found mutated in over 25% of human cancers. The processes around the RAS gene products are also involved in a variety of rare human developmental disorders called the RASopathies. RAS proteins are absolutely central regulators of growth and oncogenesis and, in turn, every regulator of RAS is poised to be fundamentally important for cancer and a broad variety of human diseases.
Driven by the interest in identifying underlying genetic determinants of drug response in a specific type of cancer of the hematopoietic system, CeMM now reports on the mechanistic link between the LZTR1 gene, previously associated with a variety of rare disorders and rare cancers, and RAS. These findings provide a new key regulator of a pathway that is one of the best studied signaling pathways in biology. As such, it represents a major advancement. The study not only sheds new light and details on the regulation of a central growth-promoting protein, but also offers a molecular explanation for an unusually large number of pathological conditions, ranging from different types of brain and pediatric cancers to developmental pathologies like Noonan syndrome.
The research team found that the protein called LZTR1, in concert with its copartner cullin 3, regulates RAS by attaching to it a small molecular tag, called ubiquitin. The modified RAS proteins demonstrate altered localization within the cell and reduced abundance. Mutational defects or inactivation of LZTR1 lead to an increase of RAS dependent pathways causing dysregulation of growth and differentiation. LZTR1 can therefore be considered a breaker of RAS action.
Publication
Johannes W. Bigenzahn, Giovanna M. Collu, Felix Kartnig, Melanie Pieraks, Gregory I. Vladimer, Leonhard X. Heinz, Vitaly Sedlyarov, Fiorella Schischlik, Astrid Fauster, Manuele Rebsamen, Katja Parapatics, Vincent A. Blomen, André C. Müller, Georg E. Winter, Robert Kralovics, Thijn R. Brummelkamp, Marek Mlodzik, Giulio Superti-Furga.
LZTR1 is a regulator of RAS ubiquitination and signaling.
Science. 2018 November 15. doi:10.1126/science.aap8210.
Funding
The study was supported by the following funding agencies and grants: Austrian Academy of Sciences, European Research Council (ERC) grants (i-FIVE 250179 and Game of Gates 695214) and starting grant (ERC-2012-StG 309634), Austrian Science Fund grant (FWF SFB F4711 and F4702), EMBO (ALTF 1346-2011, 1543-2012), NIH grants R01 EY013256 and GM102811, Cancer Genomics Center (CGC.nl), KWF grant NKI 2015-7609.
July 03, 2018
RESOLUTE: 13 academic and industry partners join forces to unlock the solute carrier class of transporters for effective new therapies
more information: https://re-solute.eu

RESOLUTE (Research empowerment on solute carriers), a public-private research partnership supported by the Innovative Medicines Initiative (IMI) with 13 partners from academia and industry, announced the start of a 5-year research project on July 1, 2018. The goal of the project is to intensify worldwide research on solute carriers (SLCs), a relatively understudied group of proteins that control essential physiological functions, and potentially establish them as a novel target class for medicine research and development.
For more information please visit the RESOLUTE website: https://re-solute.eu
The first line of immune defense against invading pathogens like bacteria are macrophages, immune cells that engulf every foreign object that crosses their way and kill it with acid, in a process called phagocytosis. In their quest to systematically study proteins that transport chemicals across cellular membranes, researchers at CeMM characterized the critical role for transporter SLC4A7 in this process, providing valuable new insights for many pathologic conditions from inflammation to cancer. Their results were published in Cell Host & Microbe.
Among the many different kinds of immune cells that patrol the body, macrophages are the first when it comes to fight against a foreign threat. With their flexible and versatile surface, they engulf every microorganism or particle that could be harmful for the health of the organism, and enclose it in an intracellular membrane vesicle called phagosome. To eliminate the threat and break it down to its constituents, the interior of the phagosome needs to be effectively and progressively acidified. For this crucial part of phagocytosis, the macrophages must undergo multiple metabolic changes, which are not yet entirely understood.
The team of Giulio Superti-Furga, Scientific Director of CeMM, in collaboration with the laboratory of Nicolas Demaurex of the University of Geneva, discovered in their latest study that a membrane protein belonging to the family of “solute carriers” (SLCs) plays an essential role in phagocytosis and phagosome acidification. Their work was published in the journal Cell Host & Microbe (DOI 10.1016/j.chom.2018.04.013).
The researchers developed an essay with special cells in which they impaired the 391 human SLC genes individually using CRISPR/Cas9 gene editing technology. Strikingly, among all SLCs, SLC4A7, a sodium bicarbonate transporter, was the only one who turned out to be essential for macrophages to undergo phagocytosis and acidification. Cells with impaired SLC4A7 were unable to acidify their phagosomes and by consequence decreased their capacity to kill bacteria.
The results of this study do not only provide new fundamental insights into the molecular functioning of one of the most important cells of the immune system. As phagocytosis plays a significant role in various pathologic conditions from inflammation to cancer, these new insights are likely of relevance beyond the context of infectious diseases. The effort to understand the role of the different cellular transporters, supported by a grant of the European Research Council (ERC), has added a small new piece to the large and fascinating puzzle coupling trafficking of chemical matter to metabolism and cellular function.
Publication
Vitaly Sedlyarov, Ruth Eichner, Enrico Girardi, Patrick Essletzbichler, Ulrich Goldmann, Paula Nunes-Hasler, Ismet Srndic, Anna Moskovskich, Leonhard X. Heinz, Felix Kartnig, Johannes W. Bigenzahn, Manuele Rebsamen, Pavel Kovarik, Nicolas Demaurex, and Giulio Superti-Furga. The Bicarbonate Transporter SLC4A7 Plays a Key Role in Macrophage Phagosome Acidification. Cell Host & Microbe, 2018. DOI: 10.1016/j.chom.2018.04.013
Funding
The study was funded by the European Research Council (ERC), the Austrian Academy of Sciences, the Austrian Science Fund (FWF), the European Commission, and the European Molecular Biology Organization (EMBO).
Acute Myeloid Leukemia (AML) is an aggressive form of blood cancer that frequently develops in children. The diseased cells often carry mutated forms of a specific gene, which is known to function within large protein networks. Researchers at CeMM and LBI-CR identified a protein of this network crucial for the survival of the cancer cells – a novel potential approach for targeted therapies. The study was published in Nature Communications.
AML is not a single disease. It is a group of leukemias that develop in the bone marrow from progenitors of specialized blood cells, the so-called myeloid cells. Rapidly growing and dividing, these aberrant cells crowd the bone marrow and bloodstream, which can be fatal within weeks or months if the disease is left untreated. Myeloid cells of various types and stages can become cancerous and cause AML, which makes the condition very heterogeneous and difficult to treat. Thus, finding drug targets that affect as many forms of AML as possible is a prime goal for researchers.
The research groups of Florian Grebien from the Ludwig Boltzmann Institute for Cancer Research, Giulio Superti-Furga, Scientific Director of the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, and Johannes Zuber, from the Institute of Molecular Pathology, tackled that question in their latest study. They were able to identify common, conserved molecular mechanisms that drive oncogenesis in the context of the large number of different MLL-fusion proteins by characterizing the protein-protein interaction networks of distantly related MLL fusion proteins. Their results were now published in Nature Communications (DOI:10.1038/s41467-018-04329-y)
The scientists identified the methyltransferase SETD2 as a critical effector of MLL-fusion proteins. Using genomic techniques including CRISPR/Cas9 genome editing, the researchers found that SETD2 loss caused induction of DNA-damage and ultimately cell death in the cancer cells. Moreover, SETD2 loss increased the lethal effect of Pinometostat, a drug that is currently in clinical development for treatment of leukemia patients with MLL fusions. These experiments might pave the way for a more effective therapy in the future using a combination of compounds.
Publication
Anna Skucha, Jessica Ebner, Johannes Schmöllerl, Mareike Roth, Thomas Eder, Adrián César-Razquin, Alexey Stukalov, Sarah Vittori, Matthias Muhar, Bin Lu, Martin Aichinger, Julian Jude , André C. Müller, Balázs Győrffy, Christopher R. Vakoc, Peter Valent, Keiryn L. Bennett, Johannes Zuber*, Giulio Superti-Furga* and Florian Grebien* (*equal contribution). MLL-fusion-driven leukemia requires SETD2 to safeguard genomic integrity. Nature Communications, 2018. DOI:10.1038/s41467-018-04329-y
Funding
The study was funded by the European Commission, the European Research Council (ERC), the Austrian Science Fund (FWF), the Austrian Research Promotion Agency (FFG), the National Institutes of Health (NIH), the National Research, Development and Innovation Office, Hungary, and Boehringer Ingelheim.
April 24, 2017
Next-Generation Microscopy with Pharmacoscopy

A novel microscopy method, developed and patented by scientists from CeMM, allows unprecedented insights into the spatial organization and direct interactions of immune cells within blood and other liquid multi-lineage tissues. The assay, called Pharmacoscopy and published in Nature Chemical Biology, is able to determine the immunomodulatory properties of drugs within large libraries on immune cells in high resolution and high throughput.
The search for new drugs, small molecule or biologicals, that influence the immune system in a desired manner is challenging: immune signaling, often a combination of communication via soluble proteins and direct interaction by cell-cell contacts, is subtle and hard to track in all its nuances. So far, there has been a lack of fast and robust technology to measure the effect of a potential immunomodulatory drug in particular in a cell-cell contact dimension.
By combining state-of-the-art high-throughput fluorescent microscopy with single cell image analysis and novel analysis algorithms, Pharmacoscopy provides a powerful solution. Developed by a group of scientists at CeMM led by Director Giulio Superti-Furga and tested in collaboration with the Medical University of Vienna, Pharmacoscopy can quantify the overall spatial patterning and direct interactions of immune cells within blood with unprecedented speed and accuracy. The method was introduced in Nature Chemical Biology (DOI:10.1038/nchembio.2360).
Combined single cell resolution and fully automated platform control, Pharmacoscopy can test large drug libraries, as available in Stefan Kubicek’s PLACEBO (Platform Austria for Chemical Biology) laboratory, for compounds with immunomodulatory potential. With this method, the scientists identified Crizotinib, an FDA approved drug for non-small cell lung cancer, to have a previously unknown immunomodulatory potential.
Publication:
Gregory I. Vladimer, Berend Snijder, Nikolaus Krall, Johannes W. Bigenzahn, Kilian V.M. Huber, Charles-Hugues Lardeau, Kumar Sanjiv, Anna Ringler, Ulrika Warpman Berglund, Monika Sabler, Oscar Lopez de la Fuente, Paul Knöbl, Stefan Kubicek, Thomas Helleday, Ulrich Jäger, and Giulio Superti-Furga. Global survey of the immunomodulatory potential of common drugs. Nature Chemical Biology, April 24, 2016. DOI:10.1038/nchembio.2360
Funding:
This study was supported by the European Research Council, the Austrian Science Fund (FWF), Swiss National Science Foundation, European Molecular Biological Organization, the Austrian Federal Ministry of Science, Research and Economy, The National Foundation for Research, Technology and Development, The Swedish Cancer Society, the Kunt and Alice Wallenberg Foundation, the Torsten and Ragnar Söderberg Foundation, and the Marie-Sklodowska Curie Fellowships.
March 07, 2017
Kickoff for Pharmacoscopy – a novel tool for precision medicine

In light of the importance of research on precision, molecular, and personalized medicine, CeMM and the Medical University of Vienna hosted on March 6, 2017 a kick off meeting to celebrate the start of Pharmacoscopy, a novel high-content screening and imaging platform to break resistance of relapsed and refractory hematological malignancies - a true bench-to-bedside circle.
This meeting presented and celebrated the collaborative project between the Superti-Furga and Kubicek laboratories at CeMM and the Division of Hematology and Hemostaseology, Department of Internal Medicine I of the medical University of Vienna. The Pharmacoscopy platform is funded with the precision medicine grant from the WWTF (Wiener Wissenschafts-, Forschungs- und Technologiefonds / Vienna Science and Technology Fund) awarded to Giulio Superti-Furga and Ulrich Jäger.
The meeting began by reviewing the importance of the strong CeMM and MedUni Wien collaborative atmosphere that has propelled basic and translational science, as reiterated by the Vice Rector for Research and Innovation Michaela Fritz. Christoph Zelinski, Director of the Department of Internal Medicine I, touched upon ongoing precision medicine programs in the MedUni Wien such as the EXACT trial. Ulrich Jäger, Head of the Division of Hematology and Hemostaseology, further spoke about the need for personalized and precision medicine in the hematological space, where functional testing that will be used to meet the aims of the WWTF grant can synergize with genetic testing that is clinically routine.
CeMM scientific Director Giulio Superti-Furga and his Senior Postdoctoral Fellow Gregory Vladimer outlined the image-based screening platform that is the backbone of this program, and how the technology is currently being used for clinical utility. The meeting was finished by Ulrich Jäger presenting interim results of an ongoing clinical study aimed at describing the benefits of data generated through this collaboration for the treatment of patients.
The Pharmacoscopy project aims to break resistance of refractory blood cancers through ex vivo automated image-based analysis of drug action, and potentially drive clinical trials of already approved drugs in off-indication blood cancers. The approach provides a very concrete and actionable platform for precision medicine and the use of off-indication approved drugs for late stage hematological malignancies. The collaboration is tuned directly to unmet clinical needs of resistant blood cancer patients.
January 16, 2017
Giulio Superti-Furga new Member of Scientific Council of the European Research Council (ERC)
Giulio Superti-Furga, Scientific Director of CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences and Professor for Medical Systems Biology of the Medical University of Vienna has been appointed Member of the Scientific Council of the ERC, on the 10th anniversary of its existence, for a function period of 4 years.
The European Research Council is the most important and prestigious funding institution for basic research in any field conducted within the European Union. Excellence is the sole criterion for selection; there are neither thematic priorities, nor geographical or other quotas for funding. Perhaps the most important funding programme of the ERC is the ERC Starting Independent Research Grant, promoting early scientific independence of promising talents with 2 million Euros over a period of 5 years. It has created a very positive impact on the attractiveness of Europe as research area. But the ERC has also other programmes, such as the ERC Advanced Investigator Grant, which fosters innovation carried out by established scientists with a proven scientific track record of excellence. Having been awarded two ERC Advanced Investigator Grants over the years and two ERC Proof-of-Concept Grants, to explore the application potential of research ideas, Giulio Superti-Furga, who also acted as ERC panel member in the past, knows the ERC well and is well suited to offer his experience to the organization that this year celebrates its 10th anniversary.
The ERC is governed by the Scientific Council, consisting of eminent European scientists and scholars including Nobel Prize laureates. Members are nominated by an independent search committee and appointed by the European Commission. Since 2014 Professor Jean-Pierre Bourguignon, a renowned French mathematician, is President of the European Research Council. From 2010 to 2013 Professor Helga Nowotny, a Viennese Professor of Social Studies of Science held this prestigious position. She was also a founding member of the ERC in 2007. In 2017 the European Research Council is celebrating its 10th anniversary.
The ERC Scientific Council acts on behalf of the scientific community in Europe to promote creativity and innovative research. Giulio Superti-Furga: “It is a great honor to accept this important responsibility, which has had a tremendously positive impact on basic research in Europe. My aspiration is to contribute to a more science and innovation-friendly climate in Europe by promoting excellence in research and ensuring that politicians protect and promote the ERC as the most successful research funding scheme of the EU. Results from basic research accompany us at every step and should therefore become a core theme in everyday life - in education, in the media and in public discussions. Society and politics must have the courage to invest in new projects, to keep pace with scientific developments and associated implications. It is important to understand science as a fundamental component of our culture and of our future and a motor for innovation and competiveness also for the European industry."
March 18, 2016
2nd ERC Advanced Investigator Grant

Giulio Superti-Furga, scientific director at the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences and Professor of Medical Systems Biology at the Medical University of Vienna is to receive the Advanced Investigator Grant of the European Research Council ERC in the amount of approximately 2.5 million euro. The term for the prestigious grant is 5 years.
"ERC Grants are the most important promotions for life sciences in Europe. Unfortunately, the situation is becoming increasingly competitive due to budget cuts," says Giulio Superti-Furga who has built up and is leading CeMM at the campus of the Medical University of Vienna and the Vienna General Hospital: "I am all the more delighted to receive this extraordinary distinction as it also confirms the relevance and quality of CeMM research." The goal of CeMM is to prepare the precise medicine of the future by decoding molecular causes for important diseases such as cancer, inflammation and immune disorders. Giulio Superti-Furga already received an ERC Advanced Grant in 2009 for the exploration of basic immune system mechanisms. In 2011, he also received the first ERC Proof of Concept Grant in Austria.
The molecular system biologist has been granted the present ERC funding for research into existing components in cell membranes that transport dissolved substances into cells. Previous studies have shown that these "cell gates", so-called SoLute Carrier Proteins (SLCs) perform an important task in regulating the cellular metabolism and are responsible for accepting medications. The goal of the research project with the title "Game of Gates" is to decode the previously unknown rules according to which cells open or close their gates, either permitting or preventing the entry of substances. Giulio Superti-Furga: "Thus far, SLCs were treated more or less as second-rate by the scientific community. However, we expect that insights from our study will significantly contribute to a new, fundamental understanding of cellular physiology and thus prepare the way for the development of new, targeted therapies for various illnesses."
The ERC Advanced Investigator Grant is being awarded to Giulio Superti-Furga after two ERC Starting Grants had been given to Andreas Bergthaler and Christoph Bock in the autumn of 2015. Moreover, CeMM Principal Investigator Kaan Boztug won the bid – also in the autumn of 2015 – for founding the Ludwig Boltzmann Institute for Rare and Undiagnosed Diseases. The "Vienna Research Groups for Young Investigators" grant of the Vienna Science and Technology Fund WWTF had also been awarded to the CeMM PI Jörg Menche. All of these promotions once again emphasize the excellent work being performed at the CeMM.
September 21, 2015
A “hot” approach for understanding drug action and identifying new drug targets

How do successful drugs actually work? And how can we identify new targets for drug discovery to enable the development of novel potential therapeutics? A key challenge for scientists in academia and the pharmaceutical industry is to find out how small molecules such as drugs or cellular metabolites act within a cell – who are the mediators and effectors required for a drug or metabolite to exert their effect e.g. on cell proliferation, shape etc.? Based on the simple fact that interaction between a small molecule - such as a drug - and a given target protein increases the thermal stability of that protein, scientists at CeMM have developed a new state-of-art approach to reveal the cellular proteins that are engaged by small molecules and metabolites. By refining the established cellular thermal shift assay (CETSA) approach in which cells are treated with a small molecule and then heated to measure the differential protein stability upon target engagement and combining it with modern protein mass spectrometry a broad view of small molecule-protein interactions is provided. A particular advantage of the new methodology is the fact that the assay works with intact living cells and thus reflects a more natural (“physiological”) context in which the small molecule and its target proteins interact without any interference considering compound uptake, efflux, metabolism as well as cellular compartments. A potential future application includes the use of patient material to identify patient specific bio markers to enable personalized medicine.
Publication:
Kilian V M Huber, Karin M Olek, André C Müller, Chris Soon Heng Tan, Keiryn L Bennett, Jacques Colinge, Giulio Superti-Furga. Proteome-wide drug and metabolite interaction mapping by thermal-stability profiling. Nature Methods, doi:10.1038/nmeth.3590.
Funding:
CeMM gratefully acknowledges funding from the Austrian Academy of Sciences, the European Union (FP7 259348, ASSET) and from the Austrian Science Fund (FWF F4711, MPN).





